 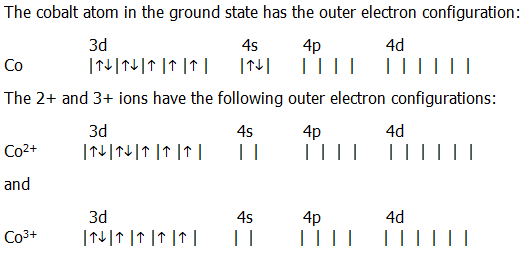
The electronic configuration of cobalt will consist of filling 27 electrons according to the Aufbau principle. We have been given cobalt which has atomic number z = 27. Potassium has nineteen electrons, one more than the noble gas argon, so. mar 2023 Electron Configuration Chart of All Elements (Full Chart) Electron. The fourth and subsequent periods follow the same pattern, except for the use of a different noble gas. Cobalt 2+ configuration Why does Co2+ have 7 electrons in the 3d orbital. Again, the number of valence electrons increases from one to eight across the third period. 
Write out the entire electron configuration for cobalt using spdf notation. Electron configuration of Cobalt (Co) Ar 3d 7 4s 2: 1s 2 2s 2 2p 6 3s 2 3p 6 3d 7 4s 2: 2, 8, 15, 2: 28. 1 shows the noble gas configurations of the third period elements. How do you write the electron configuration for Cobalt The electronic configuration. Write out the entire electron configuration for cobalt using spdf notation. The electronic configuration of Cobalt will be 1s2 2s2 2p6 3s2 3p6 3d7 4s2. 
The s, p, d, f subshells are written along with the number of the shell, like 1,2, 3, etc. Science Chemistry Cobalt, Co, has 27 electrons. The filling of electrons in various orbits is according to a principle of Aufbau that takes place from the lower energy level to the higher energy level. This page shows the electron configurations of the neutral gaseous atoms in their ground states. Consider also the electron configuration of oxygen. Then, the next electron leaves the 3d orbital and the configuration becomes: Ar4s 0 3d 6. Consider the electron configuration for carbon atoms: 1s 2 2s 2 2p 2: The two 2s electrons will occupy the same orbital, whereas the two 2p electrons will be in different orbital (and aligned the same direction) in accordance with Hunds rule. The first two to go are from the 4s orbital and Cobalt becomes:Ar4s 0 3d 7. When observing Cobalt 3+, we know that Cobalt must lose three electrons. Electronic configuration of any element consists of filling the orbital or the s, p, d, f sub – shells with electrons. The electron configuration of Cobalt is Ar4s 2 3d 7. Recall that electrons are filled from orbitals of low energy to high. Cobalt atom is Ar 3d7 4s2.The portion of Cobalt configuration that is equivalent to the noble gas of the preceding period, is abbreviated as Ar. Cobalt is a transition metal therefore, it is found in the D block of the periodic table. 
The ground state abbreviated electronic configuration of Neutral. Argon is a noble gas, which means its outer shell (3p) is filled. The Electronic configuration of any atom tells us the total number of electrons which is equal to the total number protons in that atom that is equal to the atomic number of that element. Ground State Electronic Configuration of Cobalt - neutral Cobalt atom Abbreviated electronic configuration of. The electron configuration of cobalt is 1s2 2s2 2p6 3s23p6 3d7 4s2. The electronic configuration requires filling of orbital or sub shells (s, p, d, f) according to Aufbau principle. Co (Cobalt) is an element with position number 27 in the periodic table. It is identified through atomic number, as the atomic number of an atom tells us the total number of electrons in that atom. Atomic no.Hint: The electronic configuration of any element tells us the total number of electrons present in that atom. The Shorthand electron configuration (or Noble gas configuration) as well as Full electron configuration is also mentioned in the table. Electron configuration chart of all Elements is mentioned in the table below. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
